
That is why students and professionals make a vast use of this free grams to moles calculator for accurate outputs so as to avoid any hurdle during the reaction. Each new topic we learn has symbols and problems we have never seen. Moreover, it also gives an idea about the elements that are involved in a particular chemical reaction. Chemistry Calculator Calculate chemical reactions and chemical properties step-by-step Chemical Reactions Chemical Properties full pad Examples Practice, practice, practice Math can be an intimidating subject. We convert the given mass of any compound or substance to its corresponding number of moles because it allows us to write the simplest chemical formula of the substance. If we have 5g of HCl, then we can say that it has 0.137 moles. The number of moles in 1 gram of HCl is 36.461. Let us go through the organic content below to understand how to use this free calculator.īasically, mole is the SI unit of the substance quantity.ġ mole of any compound or substance contains exactly \(6.02214076 * 10^ = 1 Mole $$ How many moles are there in 5g of an element?įor instance, let us talke an example here. 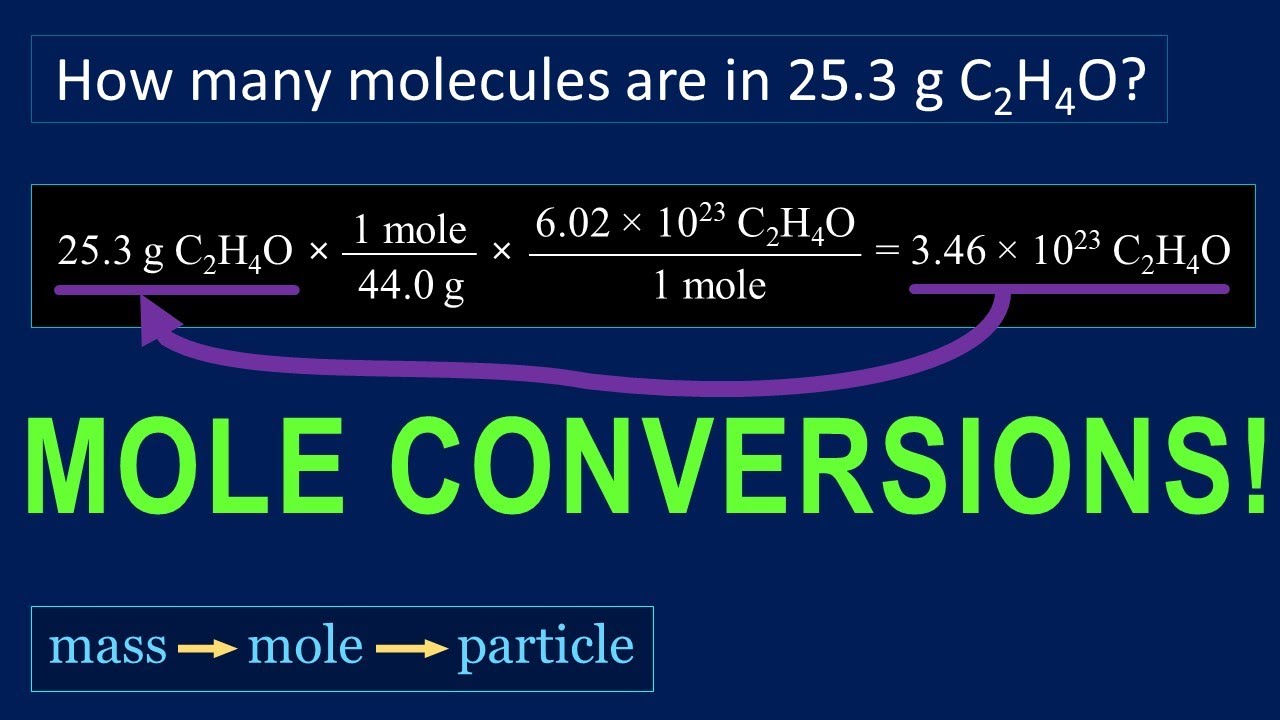
We hope this grams-to-moles calculator (or moles-to-grams calculator) will help you with your chemical calculations! You may also find our titration calculator useful.An online grams to moles calculator is exclusively programmed to convert grams to moles, moles to grams, molar mass and number of the molecules present in the substance. So we now know we need 10.8 g of NaOH to exactly neutralize our amount of hydrochloric acid. Mole × molecular weight = mass (rearrange the equation),Īs 1.626×10 23 molecules of NaOH is also equal to 0.27 moles, and we know that the molecular weight of NaOH is 40, we can use these numbers to get: Mole = mass / molecular weight (multiply both sides by molecular weight), You can also verify the results by putting the values in free grams to molecules calculator. (Mr of CO2 44) mass 44 × 0.25 11 g The calculation is the same if a substance is a metal or exists as separate atoms, but its. Carrying out grams to moles conversion: n m M. Calculate the mass of 0.25 mol of carbon dioxide molecules. Molar Mass of 5 Litres of water 5 18.015. As we need to find the mass of NaOH to add, let's quickly rearrange the equation: Now we have: Molar Mass of 1 Litre of water 18.015. The answer is rounded to three significant figures since the fewest significant figures in the calculation is three. Since there are 200.59 g Hg in 1 mol Hg, 0.0114 g Hg must be a very small fraction of a mole. Now we know the number of molecules of HCl we have, and since the reaction is 1:1, we need the exact same number of molecules of NaOH to neutralize it. 0.0114 g H g × 1 m o l H g 200.59 g H g 5.68 × 10 5 m o l H g. We can work out the number of molecules by multiplying the moles by Avogadro's constant above. Let's plug these numbers into the above equation: We know we have 10 g of HCl, which has a molecular weight of 36.5 g/mol. Let's do a quick example to help explain how to convert from moles to grams or grams to moles. If you wanted to find the concentration of the hydrochloric acid, you could use our concentration calculator. By using this moles to grams calculator, you can convert moles to mass and grams, grams to mass and moles, and last but not least mass to moles and grams. To know how to calculate moles, the equation is: 
It is, therefore, useful to find out exactly how many molecules of HCl are in the solution. It also recalculates grams per ml to moles. 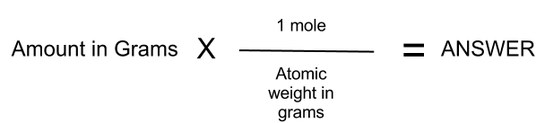
Now, you want the resulting solution to be perfectly neutral, so you don't want to add too much or too little NaOH, making it too basic or acidic respectively. Molarity Calculator This calculator converts the mass concentration of any solution into molar concentration. Let's say you want to neutralise 10 g of hydrochloric acid (HCl in water) with some sodium hydroxide (NaOH). A mole of a substance or a mole of particles is defined as exactly 6.02214076×10²³ particles, which may be atoms, molecules, ions, or electrons. So why do chemists use moles, and why do you need a mole calculator? Well, as we said above, it provides a useful metric when dealing with reactions. It turns out memorising that definition for my GCSE chemistry exam was pointless. Prior to that, a mole was defined as the number of atoms in 12 grams of carbon-12 (an isotope of carbon). The official International System of Units definition is that a mole is the amount of a chemical substance that contains exactly 6.02214076×10 23 ( Avogadro's constant) atoms, molecules, ions or electrons (constitutive particles), as of 20 th May 2019. A mole is how chemists define an amount of substance, useful when dealing with many different molecules reacting at once (i.e., any reaction). Just kidding – we're sure you've never heard that joke before. A mole is a small, subterranean mammal belonging to the family Talpidae.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
